
With the growing use of wood products worldwide, the global HWP pool sequestered 335 million tons (Mt) of CO 2 equivalent per year in 2015 and is predicted to increase to 441 Mt of CO 2 equivalent per year by 2030 4, 5. 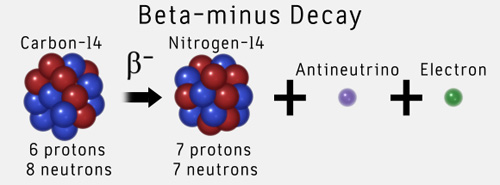
In 2020, global roundwood production was approximately 3.9 billion m 3, an increase of 25% compared to volumes in 1980 3. Carbon stored in harvested wood products (HWP) plays a mitigating role as a commodity. The differences in contributions to spatiotemporal trends between the provinces provide more specific information to make precise decisions about forest management and carbon sequestration.įorests are crucial to the global carbon cycle they absorb 7.6 ± 49 billion tons CO 2 equivalent per year, up to 30% of annual global anthropogenic CO 2 emissions 1, 2. The proportion of the HBP carbon stock in the total carbon stock increased from 20% in 2010 to 28% in 2020, indicating that bamboo products play an important role in the accumulation of carbon stocks in China. Of this, the HWP carbon stock was mainly sourced from three provinces across the north and south: Guangxi (60.8 TgC), Heilongjiang (37.2 TgC), and Fujian (24.2 TgC), and HBP carbon stock was mainly sourced from three southern provinces: Fujian (33.4 TgC), Guangxi (20.3 TgC), and Zhejiang (13.7 TgC). The results showed that China’s total carbon stocks of HWP and HBP were 328.7 teragram carbon (TgC) and 129.7 TgC between 19. Therefore, the production approach and the first-order decay method were used to estimate the spatiotemporal carbon stock change in HWP/HBP based on provincial production data from the China Forestry Statistical Yearbooks for 1987–2020. However, the contribution of HBP to carbon stocks has been neglected in mixed wood and bamboo data, especially in China. This relationship enables the determination of all values, as long as at least one is known.Harvested wood/bamboo products (HWP/HBP) constitute a large global carbon stock. 
Using the above equations, it is also possible for a relationship to be derived between t 1/2, τ, and λ. Derivation of the Relationship Between Half-Life Constants This means that the fossil is 11,460 years old. If an archaeologist found a fossil sample that contained 25% carbon-14 in comparison to a living sample, the time of the fossil sample's death could be determined by rearranging equation 1, since N t, N 0, and t 1/2 are known. N t is the remaining quantity after time, t 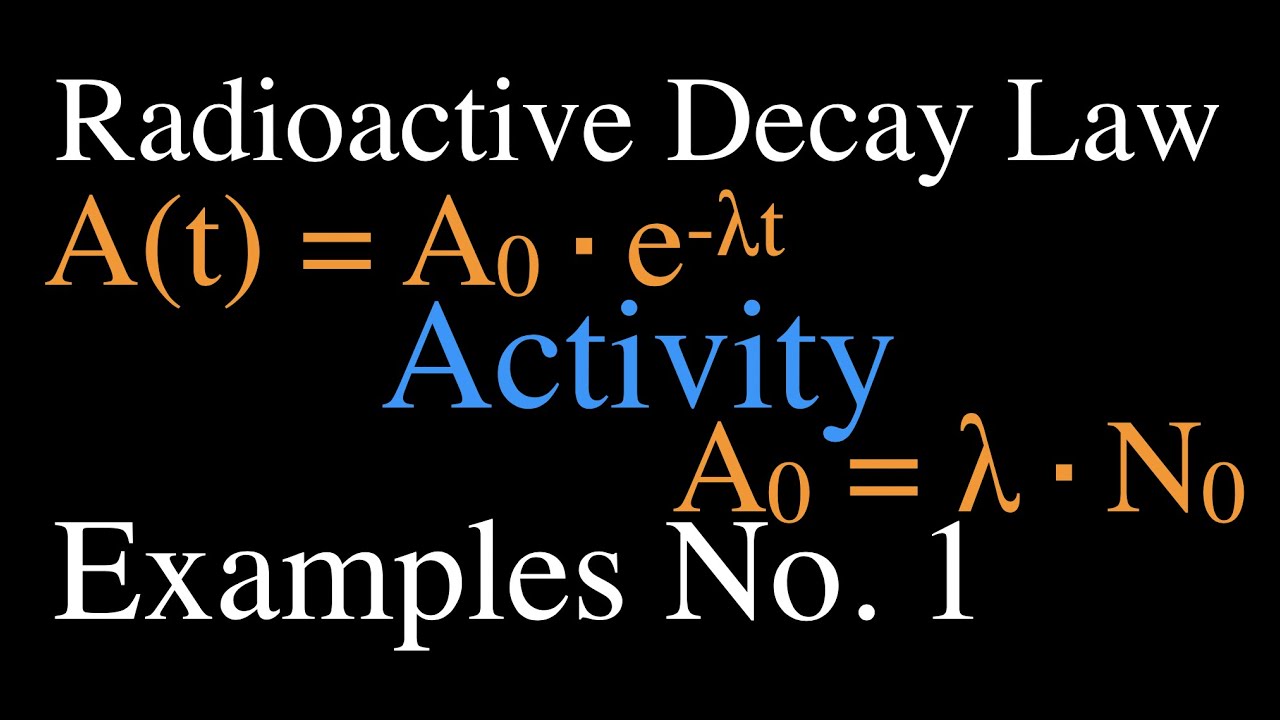
The carbon-14 undergoes radioactive decay once the plant or animal dies, and measuring the amount of carbon-14 in a sample conveys information about when the plant or animal died.īelow are shown three equivalent formulas describing exponential decay: It is incorporated into plants through photosynthesis, and then into animals when they consume plants. The process of carbon-14 dating was developed by William Libby, and is based on the fact that carbon-14 is constantly being made in the atmosphere. The half-life of carbon-14 is approximately 5,730 years, and it can be reliably used to measure dates up to around 50,000 years ago. One of the most well-known applications of half-life is carbon-14 dating. The term is most commonly used in relation to atoms undergoing radioactive decay, but can be used to describe other types of decay, whether exponential or not. Half-life is defined as the amount of time it takes a given quantity to decrease to half of its initial value.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
